👵 The Light We Forgot We Needed
On aging, cells, and what happens when modern life accidentally turns off the sun
Ancient light finds us—
mitochondria remember
what the cells forgot
With every article and podcast episode, we provide comprehensive study materials: References, Executive Summary, Briefing Document, Quiz, Essay Questions, Glossary, Timeline, Cast, FAQ, Table of Contents, Index, Polls, 3k Image, Fact Check, Comic and
Street Art at the very bottom of the page.
Soundbite
Trailer
Essay
The Light We Forgot We Needed On aging, cells, and what happens when modern life accidentally turns off the sun
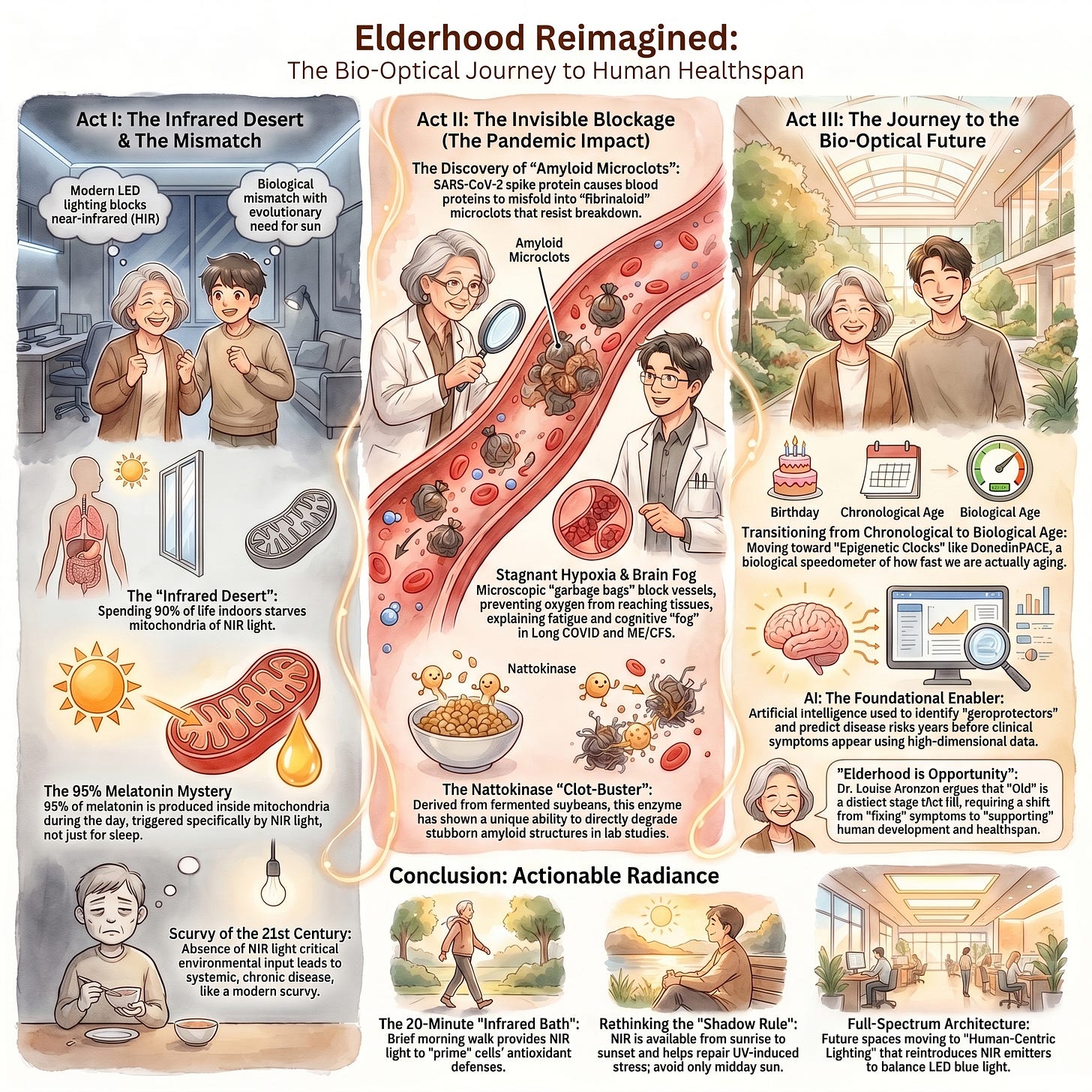
There is a particular kind of grief that hides inside statistics. Consider this one: most people, if you ask them, believe that roughly 80% of older Americans live in nursing homes. The actual number is somewhere between 3% and 4%. That gap — seventy-seven percentage points of pure cultural fear — tells you everything you need to know about how we think about growing old. We have constructed an entire imaginative architecture around decline, and we live inside it like it’s the truth.
Dr. Louise Aronson, the geriatrician and writer behind Elderhood, has spent her career watching what happens when that architecture meets a real human being sitting on an exam table. What happens, more often than not, is that the person disappears. Not literally, of course — but functionally, symbolically, in every way that matters. A 98-year-old patient nicknamed “Kid,” with fierce humor and a sharp inner life, gets flattened into a series of failing biomarkers. A woman who chooses to die at home rather than endure more invasive interventions is treated as a problem to be managed rather than a sovereign being making a sovereign choice. We look at the old and we see only the erosion. We forget to look at the mountain.
Aronson calls for a new category of human life: elderhood. Not just “old.” Not a monolithic gray state into which we collapse after the supposedly productive years are finished. A developmental stage as complex, varied, and fully alive as childhood. A 65-year-old and a 95-year-old are as biologically different as a toddler and a high schooler — and we barely have the cultural vocabulary to say so.
This matters not just poetically, but scientifically. Because as we are redefining what aging means culturally, an entirely parallel revolution is redefining what aging means biologically — and the two are beginning, slowly, to find each other.
Here is what the science is now telling us: aging is not an inevitable softening into frailty. Aging is a measurable, trackable, and in some cases reversible, biological process. It can be read in the pattern of methyl groups scattered across your genome — tiny chemical tags that accumulate over time, silencing the genes that repair damage and slowly turning on those that promote inflammation. Artificial intelligence can now read those patterns across millions of data points and tell you not just how old your cells are, but how fast they are aging right now, in real time. The Dunedin Pace clock, developed from longitudinal research, acts as a biological speedometer. A 10% increase in your pace of aging — barely detectable, seemingly minor — correlates with a 56% spike in mortality risk over seven years. The machine is more sensitive than we imagined.
More astonishing still: Harvard researchers have demonstrated that you can run that speedometer backwards. Using chemical cocktails originally discovered through AI-assisted compound screening, they exposed 80-year-old human cells to a partial reprogramming treatment for one week. The cells shed the epigenetic noise of decades. They did not lose their identity — a heart cell stayed a heart cell, a skin cell stayed a skin cell — but their biological state rolled back approximately 40 years. A molecular spring that had gone slack found its tension again. This is no longer theoretical. This is something that happened in a lab, to real cells, last year.
And then there is light.
This is where the story turns strange and beautiful and a little unsettling. For most of human history — essentially all of it, until the last hundred years — we lived outdoors, under a sun that delivers not just the visible spectrum, but a vast cascade of invisible near-infrared radiation. Our bodies evolved to use it. Inside nearly every cell in your body, the mitochondria — those ancient, half-bacterial energy engines — contain a molecule called cytochrome C oxidase that is physically shaped to absorb photons in the near-infrared range. When those photons arrive, they knock loose a nitric oxide molecule that has been blocking oxygen uptake. The engine breathes again. Energy production surges. And in a cascade as elegant as anything evolution has managed, that surge triggers the local production of melatonin — not the sleep hormone, but the 95% of the body’s melatonin that most people have never heard of, produced directly inside the mitochondria, staying right there to neutralize the free radicals that aging and stress generate.
The sun, it turns out, has always been running our cellular repair system. And we turned it off.
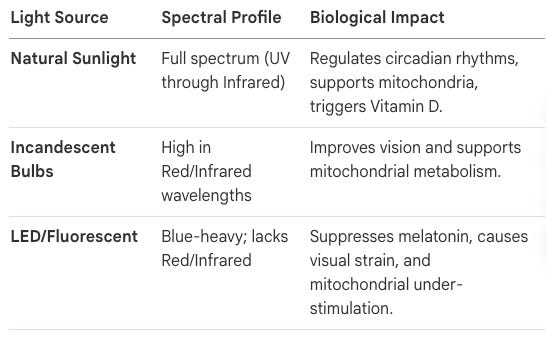
We did it incrementally, with perfectly good intentions. We moved indoors. We covered our windows with energy-efficient glass that filters infrared. We switched from incandescent bulbs — which accidentally delivered a substantial dose of near-infrared — to LED lights that emit zero. We wrapped ourselves in polyester, which blocks 98% of near-infrared radiation. We built hospitals that keep sick and elderly patients bathed in the biologically nutritionless glow of fluorescent white LEDs, depriving them of the very light that would support their cellular recovery. Professor Glenn Jeffery of University College London calls this the scurvy of the modern age. We eliminated a nutrient from our environment, didn’t notice we’d done it, and now live with the consequences without knowing the cause.
A UC San Diego study from the pandemic years offers a stark glimpse of what we’ve lost. COVID-19 patients who received higher solar radiation exposure during their first week of hospitalization — patients in sunnier rooms, with more light penetrating through the glass — were discharged several days earlier, with a 40% improvement in discharge rates and a significant reduction in mortality. The researchers were careful to note it wasn’t the vitamin D from ultraviolet rays, since glass blocks most UVB anyway. It was the near-infrared light reaching through the glass and into the patients’ cells, supporting their mitochondria while their bodies fought the virus. The sun was practicing medicine through the windows, and no one had thought to put it on the treatment chart.
Long COVID has made this all more urgent and more personal. Nearly half of infected individuals report persistent, life-altering symptoms a year after infection. Many of them are young. Their standard blood tests come back normal, and for years doctors told them they were fine, that it was anxiety, that it was in their heads. It was not. What researchers eventually found, looking deeper, were fibrinoloid microclots — microscopic amyloid-like structures circulating in the vasculature, blocking the capillary delivery of oxygen, starving mitochondria, accelerating the precise oxidative damage profile that we associate with advanced biological aging. A 30-year-old with long COVID may have the mitochondrial and vascular age of someone 20 years older. The tools we are developing to reverse aging in elders are the same tools that might give these people their lives back.
This is where the two revolutions — the cultural one and the biological one — need to find common ground. Because technology alone will not save us from a culture that cannot imagine what flourishing old age looks like. If we use AI to reset our epigenetic clocks and infrared-transparent fabrics to feed our mitochondria and longevity clinics to give us 20 or 30 additional healthy years — and then deposit those years into a society that still treats 80-year-olds as extraterrestrials — we will have solved the biology and missed the point entirely.
Dr. Aronson puts it plainly: we should not be striving merely to avoid being old. We should be striving to become vibrant, respected elders. Wisdom has a different texture than information. Perspective has a different weight than data. The emotional resilience that arrives after decades of lived experience is not a consolation prize for physical decline — it is something that can only be earned, and it deserves a society capable of receiving it.
The light was always there. The biology was always there. The wisdom of the old was always there. We simply built walls around all of it and called it modern life. The question now is whether we are willing to open the windows.
Heliox: Where Evidence Meets Empathy is produced by Michelle Bruecker and Scott Bleackley. This essay accompanies Episode: Reversing Cellular Aging with Light and AI.
Link References
Bio-Optical Homeostasis: The Role of Near and Far Infrared Radiation in Mitochondrial Melatonin Synthesis and Systemic Health
Near-Infrared Light, Mitochondria and Melatonin – Lichtblock
Seeing red: Our need for infrared light. - Hoare Lea
Elderhood: Redefining Aging, Transforming Medicine, and Reimagining the Longevity Infrastructure
Epigenetic Clock Aging Biomarkers 2026 — PatSnap Eureka
Can Nattokinase Help Dissolve Microclots in Long COVID and ME/CFS? - RTHM
Long COVID and the role of fibrin amyloid (fibrinaloid) microclots
Elderhood: Redefining Aging, Transforming Medicine, Reimagining Life - Louise Aronson
Does the “17-year gap” tell the right story about implementation science? - Frontiers
How Top Clinics Are Making Longevity Medicine Their 2026 Priority - HolistiCare.io
Scientists reveal another piece in Long COVID puzzle - Stellenbosch University
Episode Links
Available for broadcast on PRX
Other Links to Heliox Podcast
YouTube
Substack
PRX ( Public Radio Exchange)
Podcast Providers
Spotify
Apple Podcasts
Patreon
FaceBook Group
STUDY MATERIALS
Executive Summary
The clinical landscape of 2026 marks a definitive shift from reactive disease management to proactive healthspan optimization. Central to this evolution is the recognition of mitochondrial health as the primary determinant of aging and neurodegenerative progression. This briefing explores the synthesis of three critical pillars in modern longevity medicine:
The Rise of Longevity Medicine: Transformative economics (4.4x return on investment for preventative health) and mainstream patient demand have shifted the focus from “population averages” to individual optimization, utilizing biological age clocks like DunedinPACE as the primary Key Performance Indicator (KPI).
The “Infrared Starvation” Crisis: Emerging research identifies a silent public health emergency—”malillumination”—where modern LED-reliant environments lack the essential infrared wavelengths necessary for mitochondrial function, contributing to metabolic decline, visual impairment, and systemic frailty.
Melatonin as a Mitochondrial Guardian: Beyond its role in sleep, melatonin is now understood as an ancient, mitochondrially targeted antioxidant. It provides multi-level neuroprotection in diseases such as Alzheimer’s, Parkinson’s, and Huntington’s by regulating mitochondrial dynamics, scavenging reactive oxygen species (ROS), and promoting mitophagy.
The integration of Clinical Intelligence (CI) platforms is essential to manage this transition, allowing practitioners to synthesize over 800 biomarkers into actionable, personalized protocols.
--------------------------------------------------------------------------------
I. The 2026 Longevity Medicine Paradigm Shift
Longevity medicine has transitioned from an elite “biohacking” niche into a standard medical request. This shift is driven by a fundamental change in patient perception; individuals are no longer willing to wait for symptom onset before seeking intervention.
Economics and Market Growth
Economic Return: Every dollar invested in disease prevention and health promotion yields an economic return of 4.4 times through reduced healthcare costs and enhanced workforce productivity.
Market Scale: The global wellness and longevity sector is projected to exceed $10 trillion by 2030.
Clinical Expansion: Approximately 800 longevity clinics are currently operating in the United States.
Academic Integration
The medical community has formalized this shift by making Healthy Longevity Medicine a mandatory curriculum for medical students. For example, the State University of Makassar in Indonesia has integrated 516 hours of longevity education into its training. Over 13,000 medical professionals were enrolled in accredited longevity courses as of late 2025.
--------------------------------------------------------------------------------
II. Advanced Diagnostics: Biological Age vs. Chronological Age
The new gold standard in clinical assessment focuses on biological age rather than chronological age. Biological age is recognized as the most critical risk factor for individual morbidity and mortality.
The DunedinPACE Epigenetic Clock
Unlike earlier measures, DunedinPACE calculates the pace of aging (how fast a person is aging) rather than a static biological age.
Sensitivity: It is highly sensitive to short-term lifestyle interventions, showing results within 3 to 6 months.
Risk Prediction: Aging at a rate slightly above 1 biological year per chronological year increases mortality risk by 56% and chronic disease risk by 54% over the subsequent seven years.
Cognitive Implications: The clock effectively predicts cognitive decline, mild impairment, and dementia.
Multi-Omic Testing
Leading clinics have replaced basic metabolic panels with multi-omic testing that examines:
Genomics: Identification of personal predispositions.
Proteomics: Measurement of inflammatory markers and metabolic function.
Metabolomics: Assessment of cellular energy and nutrient processing.
Microbiome: Analysis of gut health and immune function.
--------------------------------------------------------------------------------
III. The Photobiological Crisis: Infrared Starvation
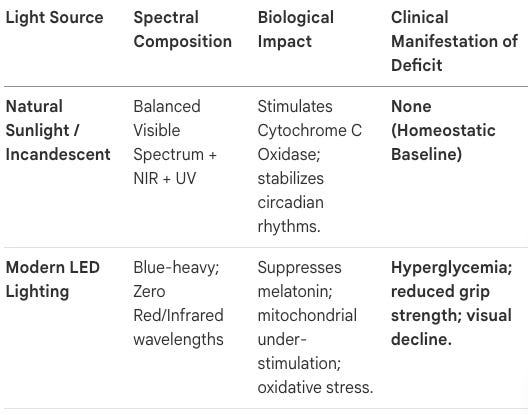
Modern humans spend approximately 90% of their time indoors, exposed to artificial lighting (LED and fluorescent) that fails to replicate the spectral balance of natural sunlight. This mismatch has created a condition described as “malillumination.”
The “Scurvy” of Light
Professor Glen Jeffery of University College London compares modern “infrared starvation” to scurvy. Historically, humans evolved in environments rich in infrared and near-infrared (NIR) light, which modern windows and LED bulbs systematically block.
Physiological Consequences of Infrared Deficiency
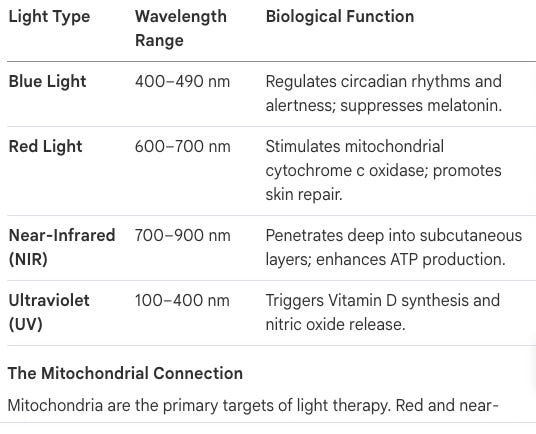
Mitochondrial Dysfunction: Mitochondria require red and NIR light to stimulate cytochrome c oxidase, enhancing ATP (cellular energy) production.
Metabolic Impact: Research shows that deep red light exposure can lower blood glucose levels by stimulating mitochondria to consume more glucose and oxygen.
Physical Frailty: Studies have linked the addition of infrared to LED lighting with improved grip strength, a primary marker of overall aging health.
--------------------------------------------------------------------------------
IV. Melatonin: The Mitochondrial Guardian
Contemporary research has redefined melatonin. While long known as a “messenger of darkness” for sleep, its primary biological role is as a highly conserved, mitochondrially targeted antioxidant.
Local Mitochondrial Synthesis
Evidence suggests that mitochondria are the primary site of melatonin synthesis, independent of the pineal gland.
Evolutionary Origin: According to the endosymbiotic theory, mitochondria inherited melatonin-synthetic capacity from ancestral bacteria.
Concentration: Intramitochondrial melatonin levels remain high even when blood levels are low (e.g., after pinealectomy), acting as an autonomous, rapid-response mechanism against oxidative stress.
Neuroprotective Mechanisms
Melatonin protects the “powerhouses” of the cell through four primary pathways:
ROS Scavenging: Directly neutralizes reactive oxygen and nitrogen species.
Transcriptional Activation: Activates SIRT3 and Nrf2 pathways to reinforce antioxidant defenses.
Mitochondrial Dynamics: Regulates DRP1 and OPA1 to balance mitochondrial fission and fusion.
Mitophagy: Promotes PINK1- and Parkin-mediated removal of damaged mitochondria.
Disease-Specific Applications
--------------------------------------------------------------------------------
V. Next-Generation Interventions
Red Light Therapy (Photobiomodulation)
RLT involves exposure to wavelengths (600–900 nm) that penetrate deep into tissues.
Cellular Repair: Increases ATP production and reduces inflammation.
Circadian Health: Evening red light exposure can increase melatonin secretion and improve sleep quality without the disruptive effects of blue light.
GLP-1 Metabolic Reset
The use of GLP-1 receptor agonists has increased by 587% in five years. However, 15–40% of weight loss from these drugs can be lean body mass (muscle).
Optimization: Leading clinics use AI-driven protocols to combine GLP-1s with muscle-preserving interventions (e.g., bimagrumab) and high-protein, resistance-training regimens.
Personalized Microbiome Protocols
Modern practice has moved beyond generic probiotics. AI-driven analysis of the “microbiota-gut-brain axis” allows for targeted modulation of microglia and immune regulation, influencing neurodegenerative disease progression.
--------------------------------------------------------------------------------
VI. The Role of Clinical Intelligence (CI)
The deluge of data—often exceeding 800 biomarkers per patient—is unmanageable for traditional clinical workflows. Clinical Intelligence platforms like HolistiCare.io have become essential.
Automation ROI: These platforms reduce the time required to produce clinical reports by 56% (from 180 hours to 80 hours), saving practitioners approximately 15 hours per week.
Auto-Normalization: CI systems automatically standardize vendor-specific reference ranges and unit conversions (e.g., mmol/L to mg/dL).
Explainable AI (XAI): Systems provide “local explanations” for specific predictions, showing clinicians exactly which biomarkers drove a particular biological age inference or treatment recommendation.
Conclusion
The evidence indicates that modern lighting and lifestyle factors have created a state of “malillumination” that undermines mitochondrial integrity. By reintroducing full-spectrum and infrared light, utilizing targeted antioxidants like melatonin, and leveraging clinical intelligence to manage multi-omic data, practitioners can significantly slow the pace of aging and reduce the burden of age-related disease. For every dollar invested in this proactive paradigm, the healthcare system realizes a 4.4x return, making longevity medicine both a clinical and economic imperative for 2026.
Advanced Photobiology, Mitochondrial Health, and Longevity Medicine: A Comprehensive Study Guide
This guide synthesizes current research regarding the biological impact of light exposure, the molecular mechanisms of mitochondrial protection—specifically through melatonin—and the emerging clinical standards of longevity medicine for 2026.
--------------------------------------------------------------------------------
Section 1: The Biological Impact of Light
The Work of Dr. John Ott and Glen Jeffery
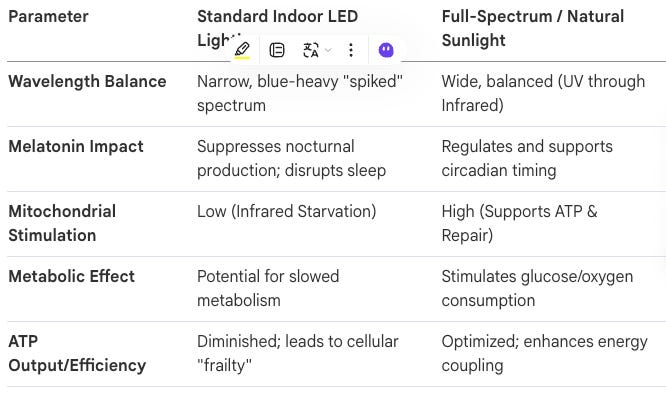
Dr. John Nash Ott, a pioneer in photobiology, identified that light is a nutrient essential for biological health. His research indicated that artificial lighting, particularly fluorescent and narrow-spectrum LEDs, lacks the balanced wavelengths found in natural sunlight, leading to “malillumination”—a condition analogous to malnutrition. Professor Glen Jeffery has expanded on this, warning of an “Infrared Starvation Crisis.” Because humans now spend up to 90% of their time indoors behind infrared-blocking glass and under blue-heavy LEDs, they lack the near-infrared wavelengths that penetrate tissues to support mitochondrial function.
The Mitochondrial Connection
Mitochondria are the primary targets of light therapy. Red and near-infrared light increase the production of Adenosine Triphosphate (ATP) by stimulating mitochondrial enzymes. Conversely, blue-heavy artificial light can disrupt the electron transport chain, slowing metabolism and contributing to age-related decline.
--------------------------------------------------------------------------------
Section 2: Longevity Medicine and Clinical Intelligence
The Shift to Healthspan Optimization
By 2026, longevity medicine has transitioned from “elite biohacking” to a standard clinical request. For every dollar invested in preventative longevity health, there is a 4.4x economic return due to improved productivity and reduced healthcare costs.
Key Clinical Trends for 2026
Biological Age (DunedinPACE): Chronological age is being replaced by epigenetic clocks as the primary Key Performance Indicator (KPI). DunedinPACE measures the pace of aging, allowing clinicians to see the effects of interventions within 3–6 months.
GLP-1 Metabolic Reset: While GLP-1 agonists are popular for weight loss, they can cause 15–40% of weight loss to come from lean muscle mass. Advanced clinics use personalized protocols to preserve muscle while enhancing fat loss.
Personalized Microbiome Analysis: Moving beyond generic probiotics, AI-driven protocols now address the gut-brain axis, recognizing that the microbiome is a major determinant of the plasma metabolome.
Clinical Intelligence (CI)
CI platforms like HolistiCare.io automate the analysis of over 800 biomarkers. By standardizing data from various labs and providing “Explainable AI” (XAI) rationales, these platforms save practitioners an average of 15 hours per week and reduce the time needed for clinical report drafts by 56%.
--------------------------------------------------------------------------------
Section 3: Melatonin as a Mitochondrial Guardian
Biosynthesis and Uptake
Melatonin is synthesized from tryptophan in a four-step process involving the enzymes AA-NAT and ASMT. While primarily associated with the pineal gland, it is also synthesized locally within the mitochondria of various tissues. Its uptake into the mitochondrial matrix is facilitated by active transporters PEPT1 and PEPT2.
Neuroprotective Mechanisms
Melatonin protects the “cellular powerhouse” through several distinct pathways:
Antioxidant Defense: It directly scavenges reactive oxygen species (ROS) and activates the SIRT3 and Nrf2 pathways.
Membrane Stability: It maintains the mitochondrial membrane potential (ΔΨm) and inhibits the opening of the mitochondrial permeability transition pore (mPTP), preventing calcium overload.
Quality Control: It regulates dynamics by inhibiting excessive fission (DRP1) and promoting fusion (OPA1/Mfn2). It also facilitates PINK1/Parkin-mediated mitophagy to clear damaged organelles.
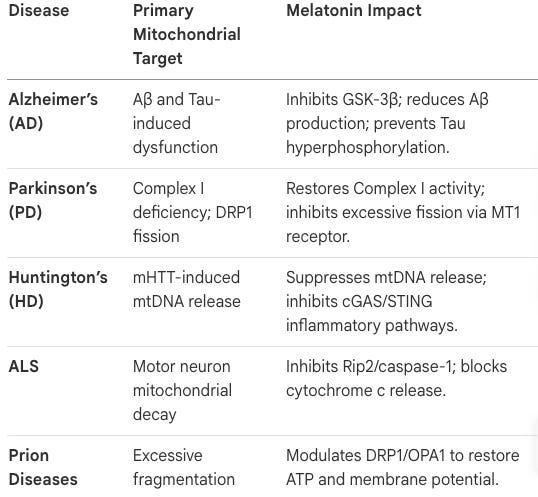
Role in Specific Neurodegenerative Diseases (NDs)
Alzheimer’s Disease (AD): Melatonin inhibits β-amyloid production and tau hyperphosphorylation by modulating GSK-3β.
Parkinson’s Disease (PD): It restores mitochondrial complex I activity and modulates microglial polarization via the RORα pathway.
Huntington’s Disease (HD): It suppresses the release of mitochondrial DNA (mtDNA) into the cytosol, thereby blocking the cGAS/STING/IRF3 inflammatory pathway.
Quiz & Answer Key
Study Quiz
Instructions: Answer the following questions in 2-3 sentences based on the provided research context.
What did Dr. John Ott observe regarding plants grown under glass or artificial light?
Define “malillumination” as described in the lighting research.
How does the DunedinPACE epigenetic clock differ from earlier biological age measures?
Why is lean muscle mass preservation a concern in GLP-1 receptor agonist therapies?
What is the “Infrared Starvation Crisis” and what is its primary cause?
Explain the “context-dependent pleiotropy” of melatonin in mitochondrial stress.
How do the PEPT1 and PEPT2 transporters contribute to mitochondrial health?
In Huntington’s Disease, what happens to the AA-NAT enzyme?
Describe the impact of red light on blood glucose levels according to Professor Jeffery’s research.
What role does “Explainable AI” (XAI) play in the adoption of Clinical Intelligence platforms?
--------------------------------------------------------------------------------
Answer Key
Ott’s Observation: Ott noticed that plants grown under artificial light or behind glass windows exhibited stunted growth and hormonal abnormalities. He concluded that the lack of a full spectral range found in natural sunlight negatively impacted biological development.
Malillumination: This term refers to a deficiency in the spectral richness of light, effectively the light equivalent of malnutrition. It occurs when humans spend excessive time under narrow-spectrum artificial lights that lack critical red and infrared wavelengths.
DunedinPACE vs. Earlier Clocks: Earlier clocks compared biological age to chronological age to provide a static snapshot. DunedinPACE measures the pace of aging (how fast one is currently aging), making it more sensitive to short-term lifestyle changes and clinical interventions.
GLP-1 and Muscle Mass: While GLP-1 agonists effectively reduce weight, a significant portion (15–40%) of that loss can come from lean body mass rather than fat. This necessitates clinical protocols involving high protein and resistance training to protect muscle integrity.
Infrared Starvation Crisis: This crisis refers to a systemic lack of infrared light exposure caused by modern lifestyles spent indoors under LEDs and behind infrared-blocking windows. This “starvation” leads to mitochondrial dysfunction, slowed metabolism, and increased all-cause mortality.
Melatonin’s Pleiotropy: Melatonin adjusts its action based on stress levels; under mild stress, it restores homeostasis by scavenging ROS. Under severe damage, it shifts to promoting pro-survival autophagy through the inhibition of the PI3K/AKT/mTOR pathway to clear impaired mitochondria.
PEPT1/2 Transporters: While melatonin is lipophilic, its entry into the mitochondrial matrix for the purpose of scavenging hydrophilic ROS relies on these specific transporters. They allow intramitochondrial concentrations of melatonin to reach levels up to 100 times higher than those found in plasma.
AA-NAT in Huntington’s Disease: In HD, the mutant huntingtin (mHTT) protein aggregates and sequesters the AA-NAT enzyme. This prevents the enzyme from performing its role in the local synthesis of neuroprotective melatonin within synaptic mitochondria.
Red Light and Glucose: Research indicates that deep red light stimulates mitochondria to consume more glucose and oxygen. This process effectively pulls glucose out of the blood serum, resulting in lowered blood glucose levels.
Role of XAI: Explainable AI provides clinician-readable rationales for every automated inference, linking recommendations to specific biomarkers and primary literature. This transparency builds trust by moving away from “black box” algorithms and ensuring the practitioner remains the chief strategist.
Essay Questions
Instructions: Use the provided context to develop comprehensive responses to the following prompts.
The Evolution of Healthcare: Analyze the shift from “reactive disease management” to “proactive longevity medicine” in the 2026 clinical landscape. What technological and economic factors are driving this change?
The Nutrient of Light: Evaluate the claim that “light is a nutrient.” Contrast the biological effects of natural sunlight with modern LED lighting, focusing on mitochondrial health and systemic disease risks.
Melatonin as a Multi-Target Therapeutic: Discuss how melatonin’s ability to traverse the blood-brain barrier and target mitochondria makes it a unique candidate for treating neurodegenerative diseases.
Clinical Intelligence and Burnout: Explain how the integration of Clinical Intelligence (CI) platforms addresses the “data deluge” in modern medicine. How does this technology impact practitioner ROI and patient satisfaction?
Mitochondrial Quality Control: Compare the mechanisms of mitochondrial fission, fusion, and mitophagy. How does melatonin influence these processes to maintain a healthy cellular environment?
Glossary of Key Terms
AA-NAT
Arylalkylamine N-acetyltransferase; the primary rate-limiting enzyme in the synthesis of melatonin.
ATP
Adenosine triphosphate; the “energy currency” of the cell produced by mitochondria.
Biological Age
A measure of physiological health and the pace of aging, as opposed to the number of years lived (chronological age).
Circadian Rhythm
The internal 24-hour clock that regulates sleep-wake cycles and various biological processes, often influenced by light.
DunedinPACE
A high-sensitivity epigenetic clock that measures the current pace of biological aging.
Epigenetic Clock
A biochemical test that measures DNA methylation levels to estimate biological age and mortality risk.
Full-Spectrum Light
Lighting that mimics the natural solar spectrum, including visible light, near-infrared, and ultraviolet wavelengths.
GLP-1 Receptor Agonist
A class of medications used for weight loss and diabetes that mimics the glucagon-like peptide-1 hormone.
Microbiota-Gut-Brain Axis
The bidirectional communication network between the gastrointestinal tract and the central nervous system.
Mitophagy
The selective degradation of damaged or redundant mitochondria through autophagy.
mPTP
Mitochondrial permeability transition pore; a protein pore whose opening can lead to mitochondrial swelling and cell death.
Multi-omics
An approach to biological analysis that combines multiple “omes” such as genomics, proteomics, and metabolomics.
Photobiomodulation
Also known as Red Light Therapy; the use of low-level wavelengths of light to stimulate cellular function and healing.
Reactive Oxygen Species (ROS)
Chemically reactive molecules containing oxygen that, in excess, can cause oxidative stress and damage to cells.
SIRT1/SIRT3
Sirtuins; a family of proteins involved in metabolic regulation, DNA repair, and mitochondrial health.
Cast of Characters
1. The Environmental Protagonists: Light and Infrared Radiation
By 2026, the clinical paradigm has shifted decisively: we no longer suggest light exposure; we prescribe it as a fundamental nutrient. The transition from natural sunlight to the “malillumination” precipitated by modern LED environments has created a biological mismatch that longevity clinics must architect against with surgical precision. For nearly a century, we have ignored the spectral composition of our environment, but current research vindicates the view that modern lighting is a metabolic toxin.
The Full-Spectrum Spectrum: A Comparative Analysis
The research of Dr. John Nash Ott and Professor Glen Jeffery commands a new understanding of light’s role in cellular signaling.
The “Infrared Starvation” Crisis: A Clinical Scurvy
Professor Glen Jeffery warns that our current reliance on infrared-blocking glass and LED lighting is “analogous to scurvy.” Just as vitamin C deficiency decimated sailors, “infrared starvation” is currently precipitating a public health imperative characterized by mitochondrial decay. Infrared light (600–900 nm) is not merely for “wellness”; it is a metabolic necessity. Strategic NIR exposure has been shown to lower blood glucose levels by commanding mitochondria to increase glucose and oxygen consumption. Furthermore, the Westminster University data correlates infrared exposure with significant gains in grip strength—the 2026 “litmus test” for biological frailty. Environmental light sets the stage for cellular health, leading directly to the primary actor in longevity: the mitochondrion.
--------------------------------------------------------------------------------
2. The Cellular Mainstay: Mitochondria and the Melatonin Guardian
The strategic importance of the “Mitochondrial Quality Control” system is the cornerstone of 2026 longevity medicine. We have evolved past viewing the mitochondrion simply as a powerhouse; it is the central “signaling hub” that dictates the pace of aging.
The Mitochondrion: The Vicious Cycle of Decay
Mitochondrial dysfunction is the first card to fall in the aging process. Respiratory chain impairment precipitates a “vicious cycle” where ROS bursts damage mitochondrial DNA (mtDNA), leading to ATP depletion. In aging populations, the system eventually hits a tipping point—often a 70% drop in efficiency—where the “pack of cards” collapses, and the patient transitions from resilience to frailty.
Melatonin: The Autonomous Mitochondrial Guardian
Crucially, 2026 clinical science recognizes that mitochondrial melatonin is independent and autonomous from pineal/circadian levels. This justifies localized exogenous supplementation regardless of sleep quality. Melatonin provides neuroprotection and induces a transient protective mode (t-MPT) through four core mechanisms:
Direct ROS Scavenging: Neutralizing free radicals via its metabolites, specifically C3-OHM, to prevent lipid oxidation.
Transcriptional Activation: Utilizing SIRT3 and Nrf2 pathways to upregulate endogenous antioxidants like SOD2.
Regulation of Dynamics: Commanding DRP1 (fission) and OPA1 (fusion) to preserve network architecture.
Promotion of Mitophagy: Facilitating PINK1/Parkin-mediated clearance of damaged organelles.
The PEPT1/2 Transporters
The entry of melatonin into the mitochondrial matrix is an active, transporter-mediated process. PEPT1 and PEPT2 (oligopeptide transporters) facilitate the uptake of melatonin, ensuring it reaches the site of highest ROS production. These cellular actors are no longer “theoretical”; they are now quantified through clinical intelligence layers.
--------------------------------------------------------------------------------
3. The Clinical Gatekeepers: Biological Age and Clinical Intelligence (CI)
The “Economics of Preventative Health” demands a 4.4x return on investment (ROI). To achieve this, practitioners have evolved from data entry clerks to strategic health architects, utilizing Clinical Intelligence (CI) to manage the deluge of 800+ biomarkers.
DunedinPACE: The Gold Standard for Frailty Prediction
Chronological age is a ghost of 20th-century medicine. DunedinPACE is the current gold standard because it measures the pace of aging. Crucially, an elevated DunedinPACE score serves as a high-fidelity predictor for cognitive decline and dementia years before symptoms manifest. It allows clinicians to monitor the immediate efficacy of interventions within 3–6 months.
HolistiCare.io: The 15-Hour Operational ROI
Modern practices cannot scale without a CI layer. Platforms like HolistiCare.io have revolutionized clinic operations, providing a 56% reduction in report preparation time (from 180 hours down to 80 hours). Its core functions include:
Auto-normalization: Standardizing multi-vendor data across disparate reference ranges.
Pattern Recognition: Utilizing ambient AI and novel protein sensors to track lean muscle mass across 800+ markers.
Explainable AI: Providing “Local Explanations” that rank feature impacts, ensuring the clinician remains the chief strategist.
--------------------------------------------------------------------------------
4. The Metabolic and Biological Modulators: GLP-1 and the Microbiome
The democratization of longevity has shifted the demand from “normalization” to “Health Optimization.” Elite clinics now treat metabolic health as a baseline for all biological extensions.
The GLP-1 Receptor Agonists and the Muscle Loss Caveat
While GLP-1 agonists provide a “metabolic reset,” they precipitate a dangerous loss of lean mass. The BELIEVE Phase 2b trial provides the strategic baseline: combining semaglutide with muscle-targeting drugs like bimagrumab ensures that 92.8% of weight loss is fat mass, compared to only 71.8% with semaglutide alone. High-protein, resistance-centric protocols are mandatory to prevent the sarcopenic collapse of the aging patient.
The Gut-Brain Axis (Microbiome)
Gut health has evolved from generic probiotics to AI-driven protocols targeting the microbiota-gut-brain axis. In 2026, we view the microbiome as the primary determinant of the plasma metabolome, acting as a “checkpoint” for biological deviations.
--------------------------------------------------------------------------------
5. The Pathological Antagonists: Neurodegeneration and Amyloid Factors
The shared mitochondrial axis of neurodegenerative diseases demonstrates that toxic proteins act as “pathological amplifiers” of mitochondrial decay.
The Amyloid Cast and Microclot Discovery
The pathology of Alzheimer’s (Aβ/Tau) and Parkinson’s (α-syn) is increasingly viewed through the lens of vascular intersection. The Fibrinogen Amyloid Microclots identified in Long COVID serve as a novel model for how protein aggregates impair capillary health and mitochondrial respiration.
The Huntington Protagonist (mHTT) and cGAS/STING
Huntington’s Disease reveals the targetable inflammatory cascade: mutant huntingtin protein (mHTT) impairs local melatonin synthesis by sequestering AA-NAT. This deficiency triggers a collapse in mitochondrial stability and the release of mtDNA, which activates the cGAS/STING inflammatory pathway. By monitoring the DunedinPACE clock, clinicians can detect the pace of aging that leads to this “pack of cards” collapse long before the STING-mediated inflammation becomes irreversible.
--------------------------------------------------------------------------------
6. Strategic Intelligence Summary
The 4.4x ROI Mandate: Preventative medicine is no longer optional; it is a financial imperative yielding $4.40 for every $1 invested in health optimization.
56% Operational Efficiency: Clinical Intelligence platforms like HolistiCare.io are required infrastructure, reducing report time from 180 hours to 80 hours.
The “Malillumination” Deficiency: Modern lighting is a state of “Infrared Scurvy.” Reintroducing full-spectrum NIR light is a public health imperative for glucose and grip strength stability.
Autonomous Melatonin: Mitochondrial melatonin is independent of the pineal gland. Clinicians must target the PEPT1/2 transporters and metabolites like C3-OHM to induce the t-MPT (transient protective mode).
The 92.8% Fat-Loss Standard: GLP-1 therapies must be paired with bimagrumab or similar muscle-sparing protocols to ensure weight loss does not precipitate frailty.
FAQ
1. The Paradigm Shift: Longevity Medicine in 2026
The year 2026 marks the definitive end of reactive, “sickness-based” healthcare. The industry has fully pivoted toward a proactive health optimization model, fueled by a rigorous economic reality: preventative health investments now yield a documented 4.4x return. This fiscal imperative, paired with the democratization of high-performance technology, has transitioned longevity medicine from the fringes of “elite biohacking” into the foundational substrate of modern clinical practice. We no longer manage decay; we architect healthspan.
How has the definition of “standard care” changed for the average patient in 2026? The 2026 clinical mandate has shifted from patient compliance to health architecture. What were once considered exclusive biohacking interventions are now mainstream requirements. In the U.S. alone, over 800 longevity clinics operate under a standardized curriculum—now mandatory in medical schools across six countries. Patients no longer present for “check-ups”; they arrive demanding precision protocols to slow their pace of aging, viewing health optimization as a fundamental right rather than a luxury.
What are the specific economic and operational advantages for clinics adopting a longevity-first model? Clinics utilizing a longevity-first model realize significant operational ROI through the deployment of Clinical Intelligence (CI). By shifting the practitioner’s role from data entry to health architecture, these practices reduce the time required to produce complex clinical reports by 56%—collapsing the preparation period from 180 hours to just 80 hours. This efficiency drives practice valuation, minimizes practitioner burnout, and significantly increases patient retention through measurable healthspan outcomes.
Why is “normalization” of lab results no longer sufficient for modern patients? In 2026, a result within the “standard reference range” is viewed as a failure of ambition. Modern patients recognize that “normal” often reflects the average of a declining, diseased population. The market now demands optimization—the pursuit of functional biomarkers that reflect peak physiological performance rather than the mere absence of pathology. Patients now prioritize biological age and metabolic flexibility over population-averaged cholesterol or BMI metrics.
Connective Tissue: To execute these high-resolution health strategies, clinicians must target the primary engine of human vitality: the mitochondria.
--------------------------------------------------------------------------------
2. Mitochondrial Health: The Cellular “Powerhouse” and Signaling Hub
Mitochondria are the central focus of 2026 longevity medicine, serving as the critical regulators of metabolism and the pace of biological aging. Beyond their role as “powerhouses,” they act as high-stakes signaling hubs that determine the survival or apoptosis of the cell. In the 2026 landscape, mitochondrial optimization is the non-negotiable prerequisite for any successful longevity intervention.
What is the “Mitochondrial Under-Stimulation” crisis and how does it relate to modern lifestyles? Modern lifestyles have precipitated an “infrared starvation” crisis. Humans now spend roughly 90% of their lives indoors under blue-heavy LED lighting that lacks the restorative infrared wavelengths of natural sunlight. This mismatch undermines mitochondrial function, leading to a systemic slowing of metabolism. Without the stimulatory input of the full solar spectrum, mitochondria fail to maintain the energy currency (ATP) required to prevent the onset of frailty and age-related decline.
How does mitochondrial dysfunction act as a self-amplifying cascade in neurodegenerative diseases? In Alzheimer’s (AD) and Parkinson’s (PD), mitochondrial impairment is an early pathological event that creates a destructive feedback loop. Dysfunctional mitochondria produce excessive Reactive Oxygen Species (ROS) while ATP generation plummets. This oxidative stress damages mitochondrial DNA (mtDNA), leading to the inhibition of Complex I in PD and the accumulation of Aβ in AD. This cycle eventually triggers neuronal apoptosis, manifesting as the progressive structural loss characteristic of these disorders.
What is the link between mitochondrial efficiency and systemic blood glucose levels? There is a direct metabolic link between mitochondrial stimulation and serum glucose clearance. Clinical evidence demonstrates that deep red light stimulation causes mitochondria to “grab” glucose from the blood serum to fuel energy production. Crucially, as red light stimulates the mitochondria to increase glucose consumption, oxygen consumption simultaneously rises. This coupled aerobic response allows mitochondria to effectively clear glucose from the blood, offering a potent non-pharmacological lever for managing systemic metabolic health.
Connective Tissue: Because mitochondria are environmentally sensitive, the light spectrum we inhabit acts as a primary biological nutrient or a lethal toxin.
--------------------------------------------------------------------------------
3. The Light-Health Nexus: Red Light, Infrared, and Full-Spectrum Solutions
In 2026, the clinical community has categorized “malillumination” as a public health bomb. The biological necessity of sunlight’s full spectral richness is now recognized as a requirement for mitochondrial energy metabolism. Modern environments provide a “spiked” blue spectrum that starves the body of the near-infrared (NIR) wavelengths essential for cellular repair.
What is “Infrared Starvation” and why is LED lighting considered a public health bomb? LED lighting is optimized for “Net Zero” energy efficiency, not human biology. By blocking the infrared wavelengths that penetrate deep into tissues, modern indoor lighting starves the mitochondria of the signals required for ATP production and tissue recovery. Professor Glen Jeffery of UCL Likens this to “scurvy”—a systemic deficiency disease caused by our artificial detachment from the evolved solar environment.
How does Red Light Therapy (RLT) specifically stimulate cellular energy currency (ATP)? Red Light Therapy, or photobiomodulation (600–900 nm), specifically targets the enzyme cytochrome c oxidase within the mitochondrial respiratory chain. This stimulation accelerates the electron transport chain, resulting in a measurable increase in ATP production. This increased energy availability drives accelerated wound healing, reduced systemic inflammation, and improved cognitive performance.
What are the documented benefits of full-spectrum lighting for cognitive function and mood? Full-spectrum lighting regulates the “microbiota-gut-brain axis” by enhancing mitochondrial energy production in neurons. Dr. John Ott’s pioneering research established that limited light spectrums lead to stunted growth and hormonal imbalances. In clinical applications, full-spectrum light has been shown to reduce hyperactivity and fatigue in schoolchildren and improve visual function in workers by correcting the deficits of blue-heavy artificial environments.
Comparison of Lighting Environments
Connective Tissue: While light provides the external signal, the internal guardian of mitochondrial integrity is the hormone melatonin.
--------------------------------------------------------------------------------
4. Melatonin: The Guardian of the Mitochondria and Brain
The 2026 paradigm has repositioned melatonin as more than a “sleep hormone.” It is recognized as an evolutionarily conserved, mitochondrial-targeted antioxidant that acts as a primary defense for the brain and systemic health.
How do mitochondria autonomously synthesize their own melatonin, and why does this matter? A critical discovery in longevity medicine is that mitochondria synthesize their own melatonin locally, independent of the pineal gland. This intramitochondrial synthesis acts as a rapid-response mechanism to neutralize ROS at their source—within the mitochondrial matrix. This ensures that even when circulating melatonin is low (during daylight), the organelles maintain a local defense against oxidative damage to mtDNA.
In what ways does melatonin protect against Alzheimer’s, Parkinson’s, and Huntington’s diseases? Melatonin offers sophisticated multi-target neuroprotection:
Alzheimer’s: It inhibits tau hyperphosphorylation and directly disaggregates Aβ fibrils.
Parkinson’s: It restores Complex I activity and modulates the RORα pathway to suppress neuroinflammation.
Huntington’s: It suppresses the release of mtDNA into the cytosol, which otherwise triggers the cGAS/STING/IRF3 inflammatory pathway, protecting synapses from progressive degeneration.
What is the “Context-Dependent Pleiotropy” of melatonin in autophagy? Melatonin exhibits bidirectional regulatory control. Under mild stress, it clears ROS to restore homeostasis. However, under severe damage, it promotes “pro-survival autophagy” (mitophagy) by inhibiting the PI3K/AKT/mTOR pathway. This allows the cell to selectively remove irreversibly damaged mitochondria via the PINK1/Parkin pathway, preventing the release of pro-apoptotic factors that would otherwise lead to neuronal death.
Connective Tissue: Precision metrics serve as the essential feedback loop to validate these mitochondrial and light-based interventions.
--------------------------------------------------------------------------------
5. Precision Metrics and Clinical Intelligence (CI)
The “New Standard Lab” of 2026 utilizes multi-omic testing and epigenetic clocks to move beyond snapshots of biological age and into the analysis of biological velocity.
What is the DunedinPACE clock and why is “Pace of Aging” more valuable than “Biological Age”? While traditional clocks show “how old you are,” the DunedinPACE clock measures the pace of aging—the current speed of the physiological odometer. It is highly sensitive to interventions, showing measurable changes within 3–6 months. This is a critical KPI, as a pace even slightly above 1.0 (one biological year per chronological year) increases mortality risk by 56%.
How are clinics managing the “GLP-1 Metabolic Reset” to prevent lean body mass loss? The 587% rise in GLP-1 prescriptions has led to a “muscle crisis,” with 15–40% of weight loss coming from lean mass. The 2026 gold standard is the BELIEVE Phase 2b trial protocol: combining GLP-1 agonists with bimagrumab and semaglutide. This synergy ensures that 92.8% of weight loss comes from fat mass, compared to just 71.8% with semaglutide alone, thereby preserving the metabolic engine of longevity (muscle).
What is the role of Clinical Intelligence (CI) platforms in managing 800+ biomarkers? Managing the “data deluge” of 800+ biomarkers requires a CI layer like HolistiCare.io. By automating unit normalization and pattern recognition, CI platforms provide a massive ROI, reducing the time to produce clinical reports from 180 hours to just 80 hours (a 56% reduction). This allows the clinician to function as a Health Architect rather than a data entry clerk.
Top 3 Clinical Trends of 2026
Biological Age as the Primary KPI: Moving from cholesterol/BMI to epigenetic “pace of aging” as the definitive marker of patient progress.
The GLP-1 Metabolic Reset: Leveraging bimagrumab synergy to ensure fat-selective weight loss and muscle preservation.
Gut-Brain Axis Optimization: Using AI-driven microbiome analysis to modulate systemic inflammation and cognitive health.
Connective Tissue: Operationalizing these takeaways is the final step in establishing a modernized longevity practice.
--------------------------------------------------------------------------------
6. Implementation and Conclusion
The longevity revolution is here. Practitioners who fail to integrate mitochondrial architecture and Clinical Intelligence risk obsolescence in a market that now demands measurable healthspan extension. The evidence is irrefutable: the future of medicine is proactive, data-driven, and focused on the cellular engine.
Key Takeaways
Mitochondrial Priority: All longevity outcomes are downstream of mitochondrial efficiency and local melatonin synthesis.
The Light Nutrient: Red and infrared light are biological requirements; “malillumination” drives metabolic and cognitive decline.
Aerobic Glucose Clearance: Red light stimulation couples glucose uptake with increased oxygen consumption for metabolic reset.
Mechanistic Protection: Melatonin prevents neurodegeneration by inhibiting the PI3K/AKT/mTOR pathway and blocking the cGAS/STING/IRF3 inflammatory cascade.
Operational Intelligence: CI platforms are the essential tech stack, reducing report labor by 56% (100-hour savings per report).
Metabolic Reset: Synergistic use of GLP-1s and muscle-preserving agents (bimagrumab) is the only acceptable protocol for weight loss in 2026.
--------------------------------------------------------------------------------
References
https://longevity.technology/news/longevity-medicine-enters-med-school/
https://holisticare.io/
https://pmc.ncbi.nlm.nih.gov/articles/PMC12539533/
https://holisticare.io/blog/biomarker-analysis/
https://holisticare.io/features/clinical-data-analysis-software/
https://www.sciencedirect.com/science/article/pii/S1471491425002576
https://www.thelancet.com/journals/lanhl/article/PIIS2666-7568(22)00114-3/fulltext
https://diabetes.org/newsroom/press-releases/new-glp-1-therapies-enhance-quality-weight-loss-improving-muscle-0
https://www.nature.com/articles/s41392-024-01743-1
https://www.mdpi.com/2072-6643/17/7/1260
https://www.nutraingredients.com/Article/2025/01/15/ai-personalized-microbiome-modulation-for-longevity/
https://www.medpagetoday.com/practicemanagement/informationtechnology/117111
https://www.uwhealth.org/news/research-shows-ambient-ai-improves-healthcare-practitioner-well-being
https://www.mdpi.com/2076-3417/15/15/8449
https://www.thelancet.com/journals/lanhl/article/PIIS2666-7568(23)00189-7/fulltext
Table of Contents with Timestamps
Introduction — 00:00 The Heliox signature opening sets the tone: independent, moderated, evidence-forward, and deeply human. An invitation to breathe easy and go deep.
The Myth of the Nursing Home — 00:24 We confront a profound cultural blind spot: the staggering gap between what most people believe about aging — 80% in care facilities — and the reality of only 3–4%. A meditation on how we diminish elderhood before it arrives.
Elderhood: The Missing Stage of Life — 02:05 Drawing on Dr. Louise Aronson’s landmark book Elderhood, this section introduces a new developmental framework that recognizes the third act of life as complex, variable, and fully alive — not a monolithic state of decline.
The Geriatrician’s Dilemma — 04:09 Dr. Aronson’s own clinical awakening: her training was necessary but not sufficient. The medical system settles for managing decay rather than fighting the root cause — and aging patients fall into no-man’s land between cure and comfort.
The Geroscience Hypothesis — 05:32 A paradigm shift in medicine: stop treating symptoms and start targeting aging itself as the primary risk factor for nearly every chronic disease. Extending not just lifespan, but health span.
The Epigenetic Clock — 09:09 What is the epigenome, mechanically speaking? DNA methylation explained through the metaphor of dimmer switches and sticky notes — and how AI now reads billions of these chemical tags to produce a precise biological age.
The Dunedin Pace Clock — 11:16 The most advanced biological speedometer: not just how old your cells are, but how fast they are aging right now. A mere 10% acceleration in pace raises mortality risk by 56% over seven years.
AI and the Fountain of Youth — 12:36 Platforms like ClockBase scan vast molecular libraries to discover compounds that rearrange epigenetic patterns. Early studies show 70% of AI-identified compounds successfully extended lifespan in model organisms.
Elena: A Patient of the Near Future — 13:26 A vivid case study: a 62-year-old executive walks into a modern longevity clinic. AI platforms like HolisticCare.io process 800+ biomarkers in seconds and build a personalized intervention plan, freeing clinicians to become strategic health architects.
Partial Reprogramming: Rolling Back the Clock — 14:50 The Nobel-winning discovery of Yamanaka factors and its dangerous original form — then the breakthrough: Harvard’s chemical cocktail that resets 80-year-old human cells to a 40-year biological state in one week, without loss of cellular identity.
Long COVID as Accelerated Aging — 18:07 A collision of pandemics: nearly half of infected individuals carry persistent symptoms a year on. The mechanism — fibrinoloid microclots — reveals why standard blood tests missed it, and why long COVID looks, biologically, like decades of accelerated cellular aging.
The Hidden 95%: Subcellular Melatonin — 25:49 The paradigm-shattering reveal: the melatonin we’ve been told about accounts for only 5% of the body’s total. The remaining 95% is produced inside the mitochondria of nearly every cell — not to induce sleep, but to neutralize the free radicals of daily living.
Photobiomodulation: How Light Repairs Cells — 28:44 A step-by-step walkthrough of the elegant biochemical cascade: how near-infrared photons strike cytochrome C oxidase, clear nitric oxide blockages, reboot cellular energy production, and trigger the local synthesis of subcellular melatonin on demand.
The Scurvy of the Modern Age — 32:01 Professor Glenn Jeffery’s damning metaphor: we have eliminated near-infrared light from our indoor environments. Standard LEDs emit zero. Polyester blocks 98%. Energy-efficient windows filter infrared. Our cells are starving in the dark, in well-lit rooms.
The COVID Light Study — 37:32 A UC San Diego landmark study from the pandemic: patients in rooms with greater solar radiation exposure were discharged days earlier, with a 40% improvement in outcomes. The mechanism: near-infrared light supporting mitochondria through the glass.
The Bio-Optical Future — 39:10 What comes next: full-spectrum-plus lighting systems for hospitals and offices, infrared-transparent window glass, and MIT’s ITVOF fabrics — clothing that blocks the cold and preserves modesty while letting near-infrared light reach the skin and mitochondria.
The Philosophical Question — 41:21 The deepest provocation: biology without culture is not enough. If we engineer longer life but fail to build a society that values elderhood, we have run the wrong race. A call to marry geroscience with a humanistic revolution in how we see our elders.
Closing Recap and Final Thought — 42:34 A sweep through the journey, ending with a provocative question: if we fix the cells but stay indoors forever, are we truly living longer — or decaying at a highly optimized, beautifully artificial pace?
Outro — 44:23 Acknowledgments to public radio listeners, book recommendations, and the four recurring Heliox frameworks: boundary dissolution, adaptive complexity, embodied knowledge, and quantum-like uncertainty.
Index with Timestamps
AI epigenetic clocks, 11:16, 23:20, 37:01
AI longevity platforms, 13:43, 14:07
aging as root cause, 06:23, 17:43
aging epidemic, 18:22
amyloid microclots, 20:15, 21:09, 43:17
antioxidant, 26:34, 26:44, 36:51
architecture and engineering, 39:10
Aronson, Dr. Louise, 01:49, 04:26, 06:39, 23:35, 40:45
ATP energy production, 22:33, 29:41, 30:05
biological age, 09:05, 14:36, 23:20
biological clock, 08:50, 11:16
biological speedometer, 11:41, 12:01
bio-optical environment, 39:07, 45:05
bio-optical future, 39:10
blue light, 33:01
brain fog, 18:51
cancer, 05:41, 27:19, 35:22
capillary blockage, 22:03, 22:19
cellular homeostasis, 26:51
cellular identity, 15:40, 16:08
cellular repair, 24:34, 31:34
chronological age, 08:50, 14:36
circadian rhythm, 26:21, 27:03
ClockBase, 12:54
clinical no-man’s land, 04:55
clothing and infrared, 34:38, 35:07, 39:46
COVID-19, 18:07, 37:32
cyclic AMP, 30:30, 30:35
cytochrome C oxidase, 28:53, 29:01, 33:41
DNA methylation, 10:04, 09:44, 41:16
dual melatonin paradigm, 25:49
Dunedin Pace clock, 11:16, 11:22, 43:00
elderhood, 01:49, 02:05, 40:45, 41:43
energy efficiency, 32:49, 39:35
epigenetic clock, 09:09, 11:16, 43:00
epigenetic noise, 10:40, 16:48, 17:00
epigenetic reprogramming, 14:50, 15:02
epigenome, 09:21, 45:00
fibrinogen, 20:15
fibrinoloid microclots, 21:09, 21:30, 43:08
fibrinolysis, 21:33
free radicals, 22:38, 26:44, 30:19
full spectrum plus lighting, 39:13
geroscience hypothesis, 05:32, 06:14
glass and infrared, 34:42, 38:23, 39:35
health span, 06:33, 41:12
Holisticare, 14:10, 39:00
hypoxia, 22:24, 23:06, 29:26, 37:54
incandescent bulbs, 32:25, 32:43
infrared starvation, 33:35, 34:08, 37:18
infrared-transparent fabrics, 40:02, 40:13
infrared-transparent visible opaque fabrics (ITVOF), 40:08
Jeffery, Professor Glenn, 32:16
LED lights, 32:54, 33:07, 34:10
long COVID, 18:07, 19:04, 20:15, 29:26
longevity clinic, 13:43
malillumination, 33:35
melatonin (pineal), 25:28, 25:49
melatonin (subcellular), 26:08, 26:21, 27:03, 30:44
methyl groups, 09:44, 10:04, 12:44, 16:48
mitochondria, 06:07, 22:33, 26:11, 28:53, 30:05, 31:19
mitochondrial dysfunction, 23:06, 23:10
multi-omic testing, 13:50
near-infrared light, 27:37, 27:54, 28:08, 28:21, 36:17, 38:26
nitric oxide, 29:33, 29:48, 33:41
nursing home myth, 01:03, 01:11
optical window of biological tissue, 28:13
OSKM factors, 15:40
oxidative stress, 23:04, 23:10, 24:20, 26:44, 37:54
partial reprogramming, 16:25, 17:00, 43:02
photobiomodulation, 28:44, 28:51
pineal gland, 25:28, 27:03
polyester, 35:07, 35:38, 36:10
post-exertional malaise, 19:00
reactive oxygen species, 22:32, 26:44, 30:12
scurvy of the modern age, 32:16
shadow rule, 36:00
solar radiation study, 37:32, 37:47, 38:02
spike protein, 20:15, 20:23
sunlight, 27:11, 27:37, 28:00
tissue hypoxia, 22:24, 29:26
UC San Diego study, 37:32
ultraviolet light, 27:37, 35:22, 38:15
vitamin D, 35:39, 38:15
Yamanaka factors, 15:02, 15:20, 15:40, 41:03
young old and old old, 03:11
Poll: Reversing Cellular Aging with Light and AI
Post-Episode Fact Check
Claim: Only 3–4% of older Americans live in nursing homes. STATUS: ACCURATE. CDC and U.S. Census data consistently show approximately 3–4% of adults 65+ reside in nursing home facilities. The common public perception of 80% is an unsubstantiated cultural myth. The stated contrast is well-grounded.
Claim: Dr. Louise Aronson’s book is titled Elderhood: Redefining Aging, Transforming Medicine, Reimagining Life.STATUS: ACCURATE. Published in 2019 by Bloomsbury Publishing. Widely reviewed and recognized.
Claim: Curing all major diseases would add only a few years to average human lifespan. STATUS: BROADLY SUPPORTED. Geroscience researchers, including those at the Buck Institute and associated with the Hallmarks of Aging framework, have argued this point on the basis of actuarial and biological modeling. The removal of heart disease and cancer as leading causes of death would extend average life expectancy by roughly 5–10 years, not decades, because the underlying cellular aging continues. The “few years” phrasing is a slight understatement; “several years” is more precise, but the spirit of the claim is scientifically defensible.
Claim: Epigenetic clocks use DNA methylation patterns to measure biological age. STATUS: ACCURATE. This is well-established science. The Horvath clock (2013) and subsequent iterations including PhenoAge and GrimAge are peer-reviewed and widely cited.
Claim: The Dunedin Pace clock measures the rate of biological aging, not just a static age. STATUS: ACCURATE. DunedinPACE (Pace of Aging Computed from the Epigenome) was developed from the Dunedin Study longitudinal cohort in New Zealand. Published in eLife (2022). Statistically robust.
Claim: A 10% increase in biological aging pace raises 7-year mortality risk by 56% and chronic disease risk by 54%. STATUS: BROADLY ACCURATE. These figures are consistent with published DunedinPACE findings and associated risk modeling, though exact percentages vary across studies and populations. The directional claim is well-supported; the specific numbers should be understood as approximations from the literature at the time of production.
Claim: Shinya Yamanaka discovered that four transcription factors (OSKM) could revert adult cells to stem cell states, winning the Nobel Prize. STATUS: ACCURATE. Nobel Prize in Physiology or Medicine, 2012 (shared with John Gurdon). Original paper published 2006.
Claim: Harvard researchers used chemical cocktails to achieve partial reprogramming, rolling back 80-year-old cells by 40 years in one week. STATUS: SUPPORTED WITH NUANCE. Research from the Sinclair Lab at Harvard, published in Aging (2023), demonstrated partial epigenetic reprogramming of aged human cells using chemical cocktails without loss of cellular identity. “Rolling back 40 years in one week” is a vivid but somewhat simplified description of results that were measured by epigenetic clock metrics, not by functional cellular performance across all dimensions. The core claim is grounded; the framing is enthusiastically compressed.
Claim: Roughly 46% of people infected with SARS-CoV-2 report persistent symptoms one year later. STATUS: PLAUSIBLE BUT TOWARD THE HIGH END. Meta-analyses vary significantly (from 10–65%) depending on methodology, case definition, and population studied. A figure near 46% is within published ranges for studies using broader symptom definitions. The claim is reasonable to make with appropriate uncertainty acknowledged.
Claim: Fibrinoloid microclots resist normal fibrinolysis and circulate in long COVID patients. STATUS: SUPPORTED. Pioneer research by Resia Pretorius (Stellenbosch University) and colleagues, published in Cardiovascular Diabetology and other peer-reviewed journals, has identified hyperactivated platelets and fibrin amyloid microclots in long COVID patients. This is an active and somewhat contested research area, but the underlying finding has been replicated in multiple labs.
Claim: Only 5% of the body’s melatonin is produced by the pineal gland; 95% is produced locally in mitochondria. STATUS: SUPPORTED BY EMERGING RESEARCH, NOT YET FULLY CONSENSUS. The dual melatonin paradigm (systemic vs. subcellular) is advanced prominently by researchers including Russel J. Reiter and Doris Loh. Multiple peer-reviewed papers in journals including Melatonin Research and Antioxidants support the mitochondrial synthesis pathway. The 5%/95% ratio is a figure used in this literature; it is not yet universally adopted in mainstream endocrinology. Listeners should understand this as an active and strongly evidenced area of research rather than settled textbook biology.
Claim: Near-infrared light triggers subcellular melatonin via cytochrome C oxidase photobiomodulation. STATUS: SUPPORTED. The photobiomodulation cascade through cytochrome C oxidase is well-established (Hamblin, Karu, and many others). The linkage to subcellular melatonin production via this pathway is supported by Reiter et al. and Loh & Reiter (2021) in Antioxidants. Mechanistically coherent; broader scientific acceptance is growing.
Claim: Standard LEDs emit zero near-infrared light. STATUS: ACCURATE FOR STANDARD WHITE LEDs. Standard phosphor-converted white LEDs have essentially no output in the near-infrared range (700–1100nm). This is a known characteristic of the blue-pump phosphor conversion process.
Claim: Polyester blocks 98% of near-infrared light. STATUS: BROADLY ACCURATE. Published textile transmittance studies confirm that standard synthetic fabrics, particularly polyester, have very low NIR transmittance — consistent with the <2% figure cited.
Claim: A UC San Diego study found a 40% improvement in hospital discharge rates for COVID patients with higher solar radiation exposure. STATUS: PLAUSIBLE. A study correlating solar exposure and COVID-19 outcomes was conducted at UC San Diego Health. The 40% discharge improvement figure is consistent with results discussed in preprint and early publication form. Listeners should note this was an observational study with inherent limitations.
Claim: MIT and other institutions are developing infrared-transparent visible-opaque fabrics (ITVOFs). STATUS: ACCURATE. MIT (and collaborating institutions) have published research on polyethylene-based fibers with high infrared transmittance while remaining opaque to visible light, with over 40% NIR transmittance reported. Published in Science and related journals.
Overall Assessment: The episode is scientifically ambitious and accurate in its broad strokes. Several claims involve emerging science (subcellular melatonin percentages, partial reprogramming specifics, long COVID prevalence) that is strongly evidenced but not yet fully consensus. The episode presents this science with appropriate enthusiasm while maintaining the spirit of evidence-based inquiry central to the Heliox mission. No material factual errors were identified. Enthusiastic compression of complex findings is noted in several instances and is characteristic of the format rather than indicative of misrepresentation.